В чем разница между временной и постоянной жесткостью и имеет ли это значение для приготовления кофе?
Жесткость - это мера количества определенных минералов в воде. Количество и пропорция минеральных веществ в вашей пивоваренной воде может существенно повлиять на вкус вашего кофе, а также повлиять на вероятность увеличения масштаба вашей эспрессо-кофемашины, поэтому стоит попытаться понять несколько ключевых понятий.
Термин «жесткость» первоначально возник из-за воздействия на мыло содержащихся в воде минералов. Минералы в жесткая вода связываются с мылом и образуют нерастворимую «пену», затрудняя образование пены и делая мыло менее эффективным при стирке. В какой-то момент люди заметили, что если воду кипятить перед использованием, она станет менее жесткой, что облегчит стирку. Жесткость, которую можно удалить кипячением, называется временной жесткостью, а жесткость, которая остается, независимо от того, сколько вы кипятите воду, называется постоянной жесткостью.
Таким образом, определение достаточно легко запомнить, но чтобы действительно понять, что происходит, нам нужно немного знать, из каких минералов состоит вода. Каждый минерал, растворяющийся в воде, состоит из ионов, которые представляют собой электрически заряженные частицы. Твердый минерал имеет равное количество положительного и отрицательного заряда, смешанного вместе, поэтому заряды нейтрализуют друг друга. Например, поваренная соль хлорид натрия состоит из равных частей положительно заряженного натрия (Na+) и отрицательно заряженные ионы хлорида (Cl-). Когда минерал растворяется в воде, ионы расщепляются ('диссоциировать') и разложите в воде.
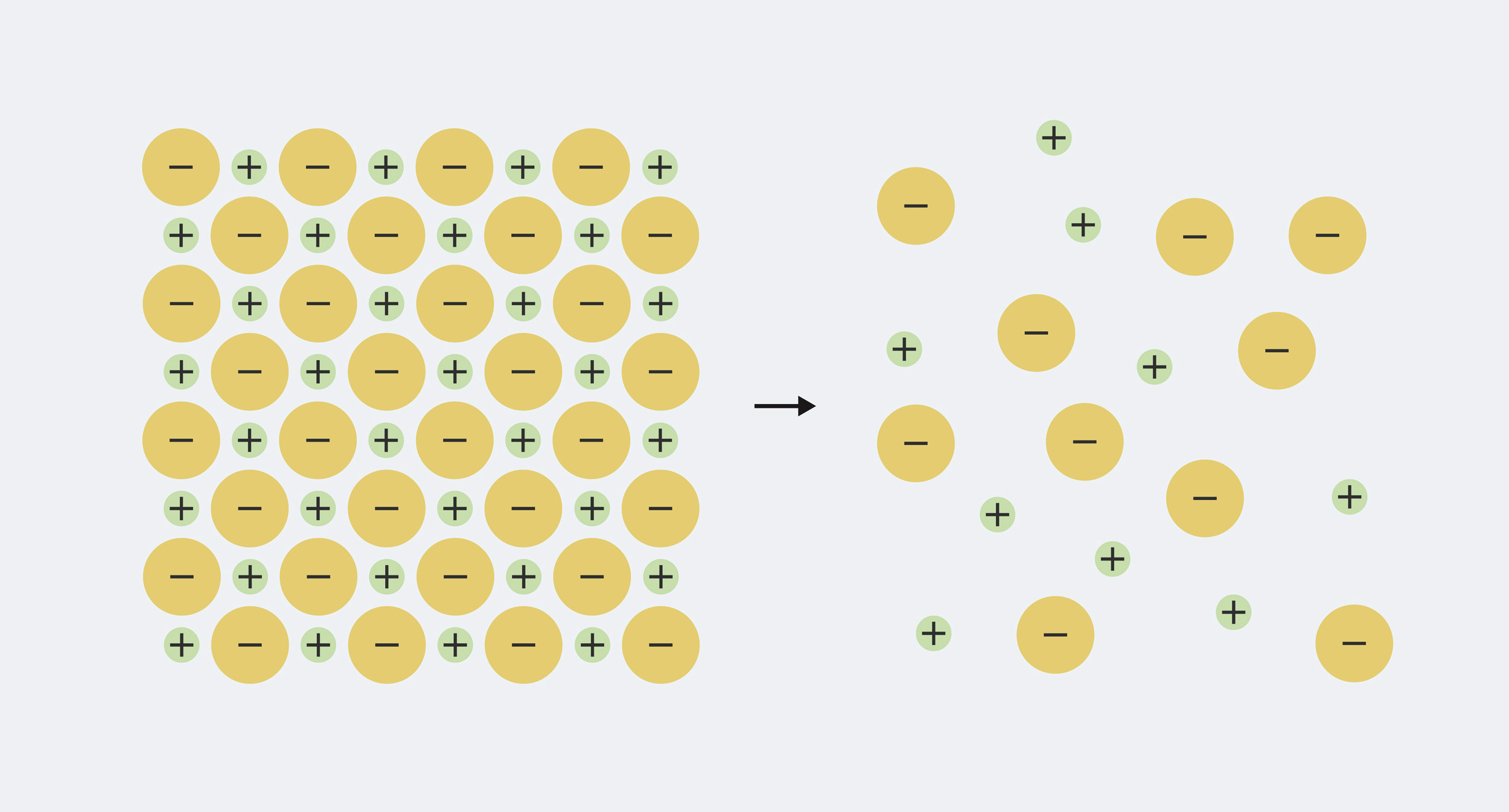 Натрий и хлорид в твердой поваренной соли плотно упакованы. Когда он растворяется в воде, он разделяется на ионы натрия (Na+) и хлорид-ионы (Cl-)
Натрий и хлорид в твердой поваренной соли плотно упакованы. Когда он растворяется в воде, он разделяется на ионы натрия (Na+) и хлорид-ионы (Cl-)
Твердость вызывается любым минералом, у которого положительно заряженный ион имеет более одного заряда. Более сильный заряд этих ионов заставляет его взаимодействовать с мылом - и это тот же заряд, который означает, что эти минералы могут помочь улучшить аромат кофе.
Кальций (Ca2+) и магний (Mg2+) составляют почти всю жесткость питьевой воды. Поскольку кальций и магний имеют два положительных заряда, они ведут себя очень похоже, поэтому их можно считать взаимозаменяемыми, когда мы говорим о твердости. Некоторые другие ионы, такие как железо (Fe3+) или алюминия (Al3+), технически может способствовать (ВОЗ 2011), но мы обычно игнорируем их, так как их количество в питьевой воде должно быть очень низким.
Таким образом, общая жесткость, также называемая «общей жесткостью» или GH, - это всего лишь мера количества положительно заряженных кальция и магния в воде. Поскольку электрический заряд в этих ионах помогает извлекать аромат молекулы, измерение GH является показателем вытяжной способности воды.
Теперь помните, что каждый положительно заряженный минеральный ион должен быть спарен с отрицательно заряженными ионами. Наиболее важным из них в воде является бикарбонат (HCO3-). Когда вы кипятите воду с бикарбонатом, он реагирует с любыми содержащимися в воде кальцием и магнием с образованием твердого вещества, называемого известковый налет (карбонат кальция и карбонат магния). Это то, что вызывает временную жесткость - кипячением воды вы формируете известковый налет, который опускается на дно. Постоянная твердость - это любая оставшаяся твердость - любой кальций или магний, у которых нет бикарбонатов, с которыми можно вступить в реакцию.
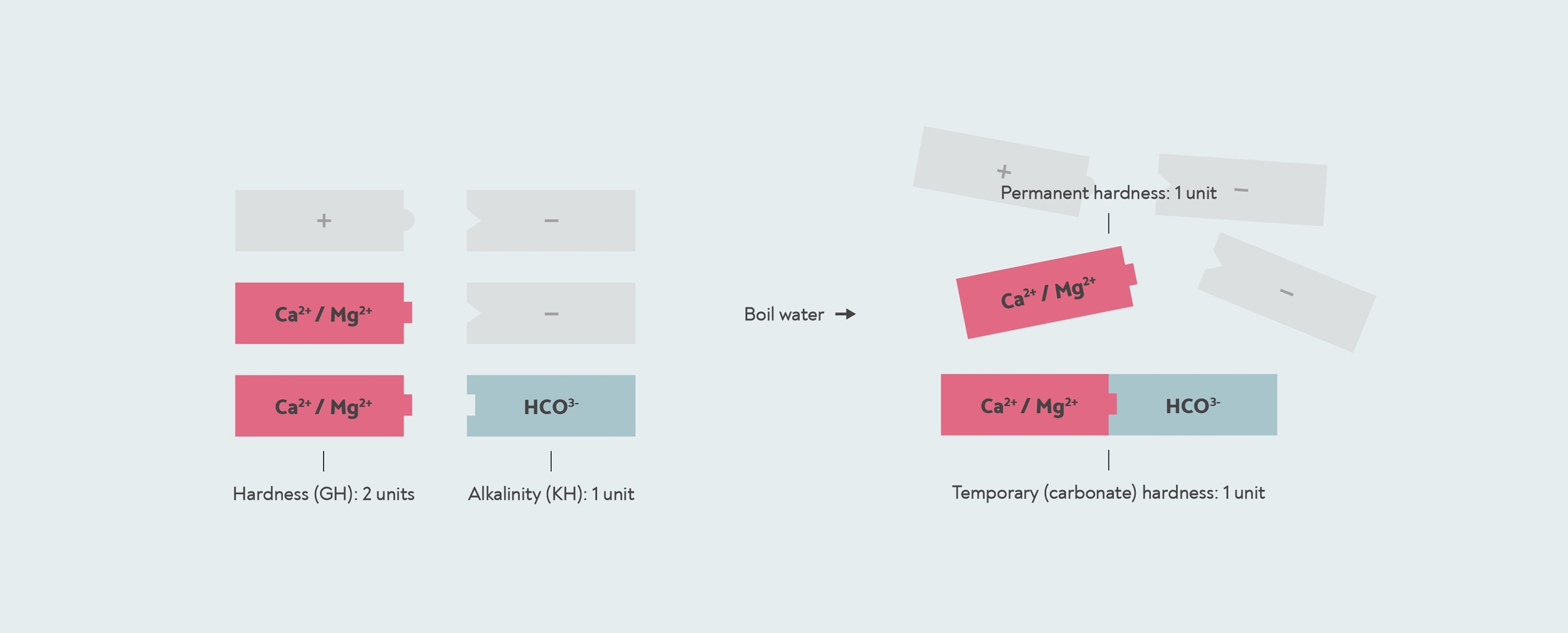 Временная и постоянная твердость. Эта вода имеет GH 2, потому что в ней есть две единицы кальция / магния (красные). Он имеет временную жесткость 1, потому что одна карбонатная единица связывается с кальцием / магнием с образованием известковый налет. Другой кальций / магний - это постоянная твердость, которая не образует известковый налет и не удаляется кипячением. Остальные положительные и отрицательные ионы минералов в воде показаны серым цветом.
Временная и постоянная твердость. Эта вода имеет GH 2, потому что в ней есть две единицы кальция / магния (красные). Он имеет временную жесткость 1, потому что одна карбонатная единица связывается с кальцием / магнием с образованием известковый налет. Другой кальций / магний - это постоянная твердость, которая не образует известковый налет и не удаляется кипячением. Остальные положительные и отрицательные ионы минералов в воде показаны серым цветом.
Когда мы измеряем жесткость воды, мы обычно делаем два измерения: GH и KH. GH - это общая жесткость, измеряющая общее количество кальция и магния в воде. KH означает Karbonathärte, что в переводе с немецкого означает карбонатная жесткость. Карбонатная жесткость точно такая же, как временная жесткость: это означает всю жесткость, которая связана с ионами бикарбоната, которые могут образовывать известковый налет.
Однако, что сбивает с толку, так это то, что типичный тест KH фактически не измеряет карбонатную жесткость. Тест на падение KH просто измеряет количество бикарбонатов в воде, также называемое щелочности. Эта мера важна еще и потому, что щелочности буферизует кислотность в кофе и делает его менее кислым. Слишком щелочности сделает вкус кофе плоским.
В нашем примере до сих пор щелочности точно такая же, как временная жесткость, потому что кальция и магния достаточно, чтобы реагировать со всем бикарбонатом и образовывать известковый налет. Так обстоит дело с большинством натуральной питьевой воды - щелочности и карбонатная жесткость или временная жесткость точно такие же, поэтому в большинстве случаев тест KH также показывает карбонатную жесткость.
Однако в некоторых случаях количество бикарбоната может превышать количество кальция или магния. Это может произойти, если вода была смягчена, или когда в питьевую воду попала соленая вода. В этом случае может быть недостаточно кальция и магния для реакции со всем бикарбонатом. В такой воде, если вы ее кипячете, вся жесткость снимается, поэтому временная жесткость такая же, как и общая жесткость, а постоянной жесткости нет вообще.
В этом случае тест KH даст вам более высокий результат, чем ваш GH. Когда результат теста KH выше, чем GH, тогда карбонатная жесткость и общая жесткость фактически одинаковы. Число, которое дает вам тест KH, на самом деле является щелочности, а не карбонатная жесткость.
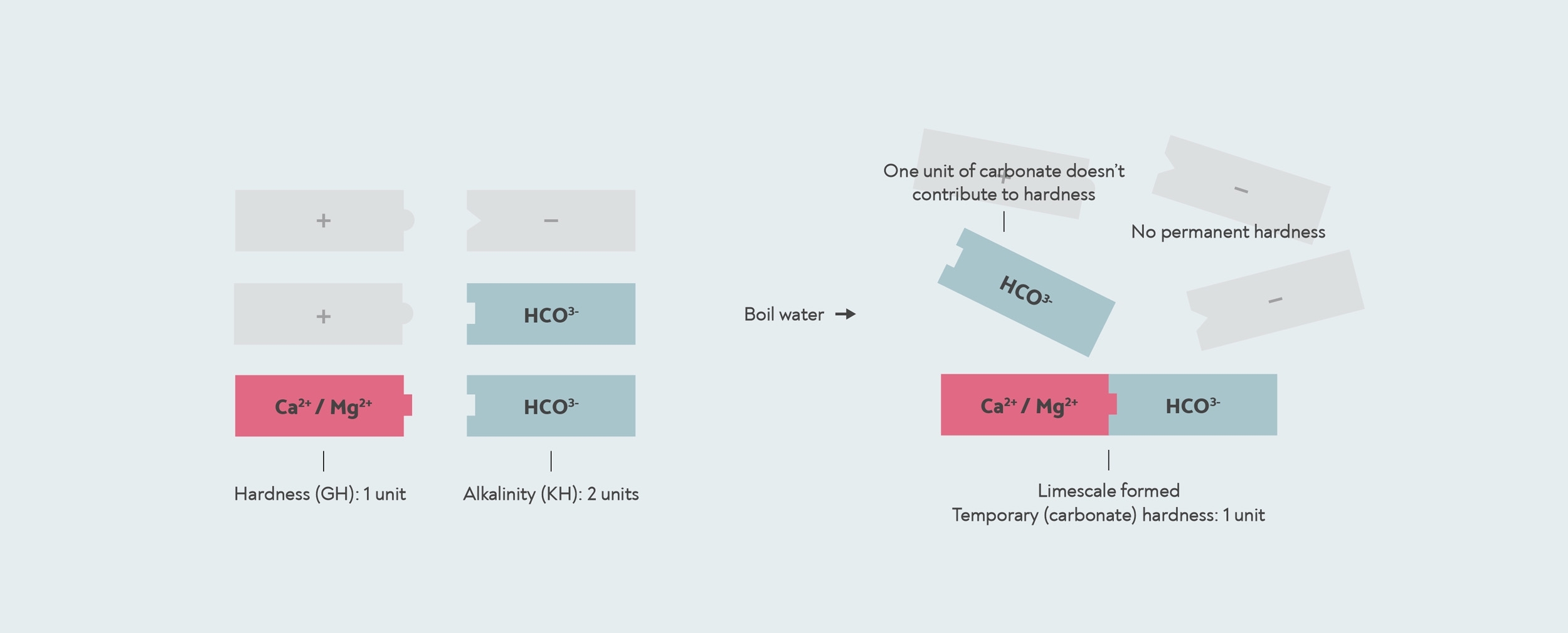 Когда щелочности выше общей жесткости, то вся жесткость временная или карбонатная. В этом случае оставшиеся щелочности не способствует твердости. Дроп-комплект KH измеряет щелочности 2, но карбонатная жесткость на самом деле всего 1, потому что для его образования требуется только 1 единица жесткости. известковый налет с участием.
Когда щелочности выше общей жесткости, то вся жесткость временная или карбонатная. В этом случае оставшиеся щелочности не способствует твердости. Дроп-комплект KH измеряет щелочности 2, но карбонатная жесткость на самом деле всего 1, потому что для его образования требуется только 1 единица жесткости. известковый налет с участием.
Однако в водопроводной воде этот случай встречается достаточно редко, поэтому большинство из нас может игнорировать его, когда проверяем нашу воду. В большинстве питьевых вод KH = щелочности = временная твердость. Постоянная твердость - это любая оставшаяся твердость, поэтому: Постоянная твердость = GH. - KH.
Таким образом, это три показателя, которые действительно имеют значение при варке кофе: общая жесткость или общая жесткость (GH), временная жесткость или карбонатная жесткость, а также щелочности. Общая жесткость помогает воде извлекать из кофе аромат. Временная твердость - это то, что определяет, сколько из этой твердости может образоваться известковый налет. Наконец, щелочности определяет, сколько воды удалит кислотность из кофе.
Если KH ниже GH, то тест KH даст вам значение как для временной твердости, так и для щелочности, потому что KH определяет, сколько GH может образовывать известковый налет. Если же KH выше GH, то KH просто измеряет щелочности, а временная твердость такая же, как и общая твердость, потому что «лишний» KH не может образовывать известковый налет сам по себе.





Основываясь на объяснении, я недоумеваю, почему существует такой отказ от ответственности при использовании рецептов воды: «Примечание: если вы сами готовите воду с солями магния (например, английской солью), то расчет LSI не применяется, поскольку нет кальций присутствует. Мы не рекомендуем использовать такую воду в кофемашинах для приготовления эспрессо». –
На этой странице упоминается, что магний связывается с присутствующими карбонатами, что приводит к KH (временной жесткости). Это приведет к легкому образованию накипи, и это хорошо, поскольку, согласно одному из ответов выше, это создает хороший цикл, при котором на котле образуется тонкий слой защитного известкового налета, который постоянно обменивается новой реминерализованной водой, добавляемой в качестве вода кипит и остывает.
Моя цель — создать воду, которая имеет прекрасный вкус, но при этом безопасна для использования в кофемашине для приготовления эспрессо. Могу ли я безопасно использовать рецепты по этому URL-адресу? https://www.baristahustle.com/app-archive-main/the-water-calculator/
Привет, Самиззл. Дело в том, что если вы создадите кофейную воду, используя дистиллированную воду, которая не содержит кальция, даже после добавления в нее английской соли и бикарбоната через концентраты, которые мы рекомендуем здесь. https://www.baristahustle.com/blog/diy-water-recipes-redux/, вполне возможно, что эта вода все еще не образует накипи, а также может привести к ржавчине частей вашего котла или других компонентов. Вот почему мы создали калькулятор бутылок, чтобы вы могли с самого начала реминерализовать воду, содержащую известное количество кальция. С course вам нужно быть осторожным, чтобы не получить слишком накипеобразующую воду, поэтому, даже если вы найдете хорошую базовую воду, которая, согласно этикетке на бутылке, может содержать, скажем, 30 миллиграммов / л кальция, будьте уверены. чтобы проверить его GH тоже.
Интересно, я обнаружил, что, хотя я и понял это, я вообще ничего не понимаю :S. Я все еще пытаюсь смириться с разницей между магнием и кальцием в отношении образования накипи, учитывая следующее утверждение: «Поскольку кальций и магний имеют два положительных заряда, они ведут себя очень похожим образом, поэтому их можно считать взаимозаменяемыми, когда мы говорим о твердости».
Вы хотите сказать, что карбонат кальция откладывает известковый налет легче, чем карбонат магния? Если да, то можно ли подсказать, какое количество кальция я мог бы добавить в свой 1 л концентрата жесткости (в дополнение к 2,45 г английской соли, которую я уже добавил), чтобы правильно сбалансировать его и избежать коррозии?
Правильно. Магний может образовывать своего рода налет, но он не имеет склонности прилипать к поверхностям так же, как известковый налет (известковый налет по определению — это кальций).
Потенциал образования накипи в вашей воде (измеренный с помощью LSI) зависит не только от количества кальция — он также зависит от того, что еще присутствует, а также от pH воды и даже температуры. Поэтому я не могу дать вам конкретную рекомендацию, но советую вам проверить LSI вашей воды с помощью нашего Калькулятор воды.
Сказав все это, является ли коррозия серьезной опасностью для вашей машины или нет, зависит от того, из чего она сделана и как она устроена. Многие машины без проблем работают на дистиллированной воде в течение многих лет, у других может возникнуть коррозия даже при использовании природной воды. Для получения дополнительной информации об этом, посетите АМА, которую мы проводили, посвящена воде.
Отличная информация, спасибо. У меня есть еще один вопрос к вам. Наша вода относительно жесткая, 350-400 частей на миллион. Мы находимся в процессе выбора либо устройства для смягчения воды, которое удаляет ионы минералов, либо кондиционера для воды TAC, который заставляет ионы кристаллизоваться, но не удаляет их. Как присутствие этих инертных кристаллов повлияет на напиток?
Привет Дэн, спасибо за комментарий. У нас нет непосредственного опыта работы с TAC или связанными с ним вариантами очистки воды (такими как магнитные или электрические кондиционеры для воды), но мы настроены скептически.
Говорят, что этот тип кондиционера для воды работает так, что заставляет формироваться микрокристаллы карбоната кальция, другими словами, крошечные кристаллы известкового налета. Теория заключается в том, что, образуя кристаллы в воде, вы предотвращаете их образование на металлической поверхности вашего водонагревателя.
Вполне возможно, что TAC может помочь уменьшить образование известкового налета в определенных ситуациях, но насколько хорошо это работает в большинстве реальных условий, далеко не доказано. Эта страница имеет хорошее представление о том, что о них мало что известно, и указывает на главный недостаток: кристаллы образуются только в том случае, если вода перенасыщена, поэтому, хотя до определенного момента она может работать, она может быть не такой эффективной, как умягчитель для получения до уровня жесткости, который нам нравится для кофе.
Как они цитируют: «Он не может предотвратить отложение накипи на высокотемпературных поверхностях, где карбонаты будут менее растворимы, и не может удалить ионы жесткости до уровня, при котором они не будут образовывать пену с мылом». Я подозреваю, что если вода все еще достаточно жесткая, чтобы образовать мыльную пену, она достаточно жесткая, чтобы оказать негативное влияние на кофе. Также стоит помнить, что эти кристаллы не являются «инертными» — это просто маленькие кристаллы известкового налета, и ничто не препятствует их повторному растворению в воде.
Если вы или кто-либо из наших читателей попробуете кондиционер TAC для своего кофе, мы будем рады узнать о вашем опыте. Но пока мы рекомендуем придерживаться умягчителей или установок обратного осмоса, чтобы быть уверенными в хороших результатах.
Я всегда считаю воду самой загадочной и сложной частью кофе. Вопрос: если большая часть водопроводной воды имеет более низкий уровень HCO-, чем Mg + и Ca +, и когда вода кипятится, все HCO- «спариваются» с Mg + и Ca + (образуя известковый налет), как может остаться какой-либо HCO- в воде? вода для снижения кислотности?
у меня такой же вопрос, как и у вас. ищу ответ от BH
Привет, Уилл - здесь есть две вещи.
Во-первых, для образования известкового налета требуется время. Если вы варите чайник для кофе, к тому времени, как вы варите кофе, образуется лишь небольшое количество известкового налета, поэтому после кипячения в воде все еще будет много бикарбоната. Вам придется долго кипятить чайник, чтобы выпал весь известковый налет.
Теперь в кофемашине эспрессо чайник кипит очень долго - навсегда, если вы его не выключите. Но в этом случае вступает в действие второй фактор - имеющийся в бойлере известковый налет также начинает растворяться в воде. Существует точка равновесия, при которой количество растворенного известкового налета совпадает с количеством образующегося - это идея, лежащая в основе индекса насыщенности Ланжелье, о котором вы можете узнать больше в Водное русло.
Ах, спасибо, в этом есть большой смысл. Таким образом, если вы держите воду при высокой температуре в «чистом» сосуде в течение длительного времени, вы можете выпустить больше бикарбоната, но, очевидно, фильтрация становится более разумным вариантом.
Привет! Меня давно озадачивает простой вопрос: если Mg образует чешуйки так же хорошо, как Ca, почему Хендон и M CD выступают за замену Mg на Ca? Или Mg как-то «безопаснее» с точки зрения масштабирования (учитывая, что 95% источников предупреждает только о Ca, это определенно кажется так…)?
Привет, спасибо, что разместили это - хороший вопрос.
Короткий ответ: они думают, что это улучшает вкус. Они предполагают, что Mg пропорционально извлекает больше желаемых ароматических соединений. Это основано на их собственном вкусовом тестировании, и они предлагают механизм для этого с помощью вычислительной химии (https://pubs.acs.org/doi/pdf/10.1021/jf501687c), но, насколько мне известно, это не было подтверждено независимыми источниками.
Основная причина, по которой большинство источников предупреждают об отложении кальция, заключается в том, что кальций обычно является основным компонентом жесткости питьевой воды, поэтому образование отложений магния не является большой проблемой. Однако замена кальция на магний также может немного уменьшить влияние накипи. Магний нарушает кристаллическую структуру отложений карбоната кальция, что увеличивает вероятность образования кристаллов арагонита, которые немного более растворимы, чем кристаллы кальцита, образованные чистым карбонатом кальция. Сам магний образует чешуйки из карбоната магния и гидроксида магния, но оба они немного более растворимы, чем гидроксид кальция при 100 ° C. Таким образом, кажется возможным, что более высокая доля магния также «безопаснее».
Ура, спасибо за ответ, это действительно полезно! Это то, с чем я еще не сталкивался.
«… Но оба они немного более растворимы, чем гидроксид кальция при 100 ° C» - вы имеете в виду CaCO3, верно?
Я живу в США, и из того, что я могу понять из отчета о качестве воды из моего местного района водоснабжения, видно, что у нас очень жесткая вода. Поэтому для приготовления кофе я использую воду в бутылках. Я наткнулся на пиковый кувшин для воды. Мне было интересно, все ли у вас была возможность это проверить.
Привет Тимоти. Фильтр Peak Water очень хорош. Скотт Рао очень рекомендовал его в своем недавнем декабрьском информационном бюллетене. Один комментарий, который мы слышали несколько раз, заключается в том, что он становится менее последовательным во времени, но это ожидается со всеми системами ионного обмена.
Я сравнил Peak с кувшином-фильтром Brita Marella (с использованием картриджей Maxtra). Они оба почти одинаково эффективны с точки зрения ионного обмена (картридж Brita немного использовался, поэтому я сделал на него скидку), но TBH Brita кажется гораздо более надежным с точки зрения материалов и дизайна. К сожалению, Peak производит впечатление пластика низкого качества, произведенного в Китае, и я боюсь, что это продлится недолго.