The Langelier Saturation Index
In 1936, Professor W. F. Langelier of the University of California, Berkeley, produced a research paper. It described his new method for calculating the pH at which water becomes saturated with calcium carbonate. This point of saturation is known as the pHs. The Langelier Saturation Index (LSI), as it came to be known, expresses the difference between the actual pH of a sample of water and the pH it would have if it were saturated in calcium carbonate.
A reading of zero suggests a water is balanced and will neither dissolve or be likely to precipitate limescale. A negative reading indicates that water is undersaturated and has the ability to dissolve calcium carbonate. A very low reading such as -2 indicates that water is very likely to dissolve calcium carbonate, and we infer from this that the water will probably cause corrosion of boilers and pipes. A positive reading indicates that water is supersaturated in calcium carbonate and will precipitate limescale. A reading of 2, for example, indicates water that is prone to leaving significant deposits of limescale.
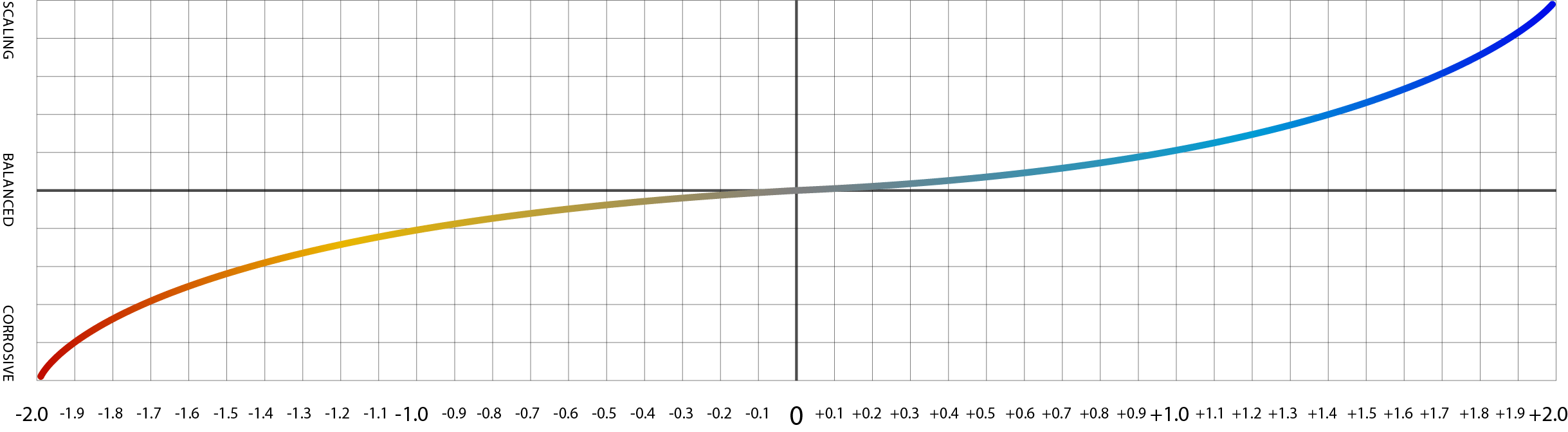
An image of the Langelier scale where negative numbers indicate corrosivity and positive numbers indicate scale-forming potential
Langelier saw the benefits of using a saturated water’s scale-forming potential strategically: His idea was to use limescale as a kind of Teflon coating. In his 1936 paper he states, ‘Calcium carbonate is the salt which is the most useful in forming, or together with rust, assisting in the formation of a self-healing or natural protective coating.’
If we take the recommendations of Scott Guglielmino (from the preceding lesson) as a guide, LSI values ranging from 0.2 to 0.7 represent a safe level of water saturation that will promote some, but not excessive, limescale formation.
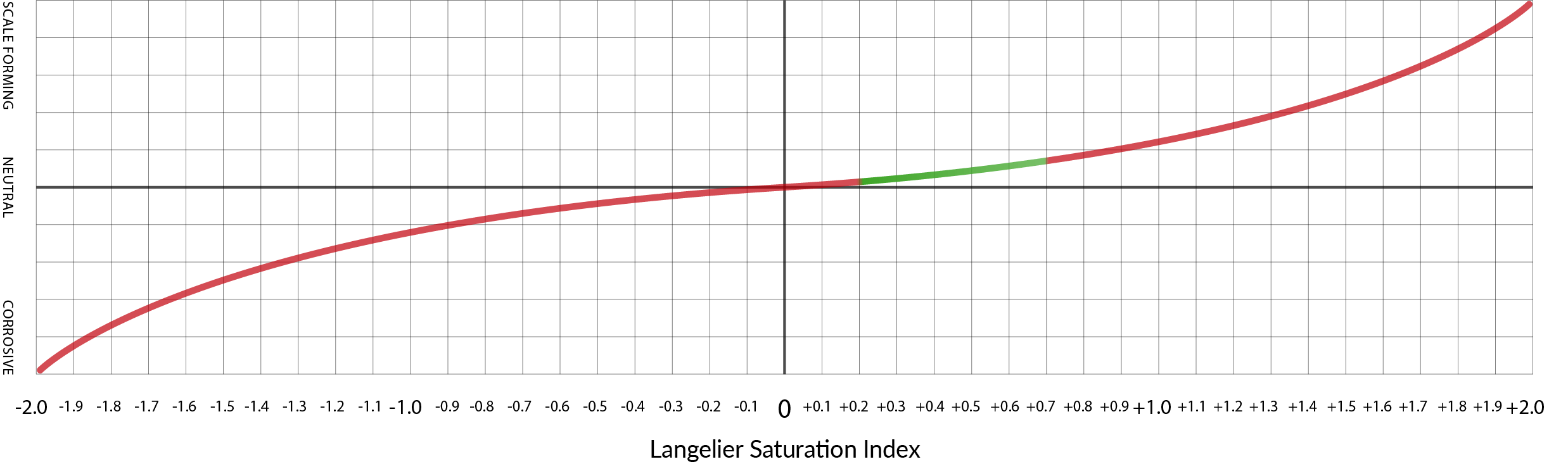
The green section of this illustration depicts La Marzocco’s safe water range with LSI values ranging from 0.2 to 0.7